10 ml x 100 mg/ml
CATEGORIES
COLLECTIONS
MANUFACTURERS
WAREHOUSES
Wholesale
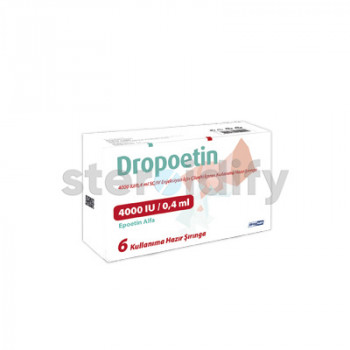
DROPOETIN 4000IU
Characteristics
Description
DROPOETIN 4000IU - DROGSAN
Dropoetin is indicated for the treatment of anaemia associated with chronic renal failure in paediatric and adult patients on haemodialysis and peritoneal dialysis.
Dropoetin is indicated for the treatment of severe anaemia of renal origin accompanied by clinical symptoms in adult patients with renal insufficiency not yet undergoing dialysis.
Dropoetin is indicated for the treatment of anaemia and reduction of transfusion requirements in adult cancer patients (with non-myeloid malignancies) receiving chemotherapy.
Dropoetin is indicated for the treatment of anaemia in adult HIV infected patients being treated with zidovudine having endogenous Dropoetin levels ≤500 mU/ml.
Dropoetin is indicated in adults to facilitate autologous blood collection within a predeposit programme and decrease the risk of receiving allogeneic blood transfusions in patients with moderate anaemia [haematocrits of 33-39%, haemoglobin of 10-13 g/dL, (6.2-8.1 mmol/l), no iron deficiency], who are scheduled for major elective surgery and are expected to require more blood than that which can be obtained through autologous blood collection techniques in the absence of Dropoetin. Treatment should only be given to patients if blood saving procedures are not available or insufficient when the scheduled major elective surgery requires a large volume of blood (4 or more units of blood for females or 5 or more units for males).
Dropoetin is indicated to augment erythropoiesis in the perisurgical period in order to reduce allogeneic blood transfusions and correct postoperative anaemia in adult non-iron deficient patients undergoing major elective orthopaedic surgery. Use should be restricted to patients with moderate anaemia (eg Hb 10-13 g/dL) who do not have an autologous predonation programme available and with expected moderate blood loss (900 to 1800 ml).
Dropoetin is indicated for the treatment of anemia in patients with Low or Intermediate 1-risk Myelodysplastic Syndrome (MDS). MDS patients considered for therapy should have a hemoglobin less than 10 g/dl.
How should I use Dropoetin?
Use Dropoetin as directed by your doctor. Check the label on the medicine for exact dosing instructions. Check the label on the medicine for exact dosing instructions.
- Dropoetin comes with an extra patient information sheet called a Medication Guide. Read it carefully. Read it again each time you get Dropoetin refilled.
- Dropoetin is usually given as an injection at your doctor's office, hospital, or clinic. If you will be using Dropoetin at home, a health care provider will teach you how to use it. Be sure you understand how to use Dropoetin. Follow the procedures you are taught when you use a dose. Contact your health care provider if you have any questions.
- When drawing up a dose into a syringe, be sure to follow the procedure demonstrated to you to prevent contamination of the vial, syringe, or medicine. Never touch the rubber stopper of the vial or the needle of the syringe with your fingers.
- Always carefully check that you have drawn up the correct dose before administration.
- Do not shake Dropoetin. Do not use Dropoetin if it has been shaken.
- Do not use Dropoetin if it has been frozen.
- Do not dilute Dropoetin. Do not mix it with other solutions unless your doctor directs you otherwise.
- If you are using a single-dose vial, throw away any medicine left in the vial after you have used your dose. Ask your doctor, pharmacist, or other health care provider if you have questions about whether you are using a single-dose vial.
- Do not use Dropoetin if it contains particles, is cloudy or discolored, or if the vial is cracked or damaged.
- Keep this product, as well as syringes and needles, out of the reach of children and pets. Do not reuse needles, syringes, or other materials. Ask your health care provider how to dispose of these materials after use. Follow all local rules for disposal.
- Continue to use Dropoetin even if you feel well. Do not miss any doses.
- If you miss a dose of Dropoetin, contact your doctor right away.
Ask your health care provider any questions you may have about how to use Dropoetin.
Dropoetin side effects
The following serious adverse reactions are discussed in greater detail in other sections of the label:
- Increased Mortality, Myocardial Infarction, Stroke, and Thromboembolism
- Increased mortality and/or increased risk of tumor progression or recurrence in Patients With Cancer
- Hypertension
- Seizures
- PRCA
- Serious allergic reactions
Customer Reviews
Customers who bought this item also bought:
Buy 10+ pcs for $11.00 each (You Save $50)
Buy 20+ pcs for $9.00 each (You Save $140)
Buy 3+ pcs for $14.00 each (You Save $6)
Buy 5+ pcs for $14.00 each (You Save $10)
Buy 10+ pcs for $8.00 each (You Save $40)
Buy 20+ pcs for $7.00 each (You Save $100)
Buy 3+ pcs for $11.00 each (You Save $3)
Buy 5+ pcs for $10.00 each (You Save $10)